

Body Protection Compound-157 (BPC-157) is a synthetic 15-amino-acid peptide derived from a naturally occurring protein in human gastric juice that has attracted widespread interest for its proposed tissue-healing and regenerative effects. Research to date is predominantly preclinical (animal and lab studies), with very limited human clinical evidence. This article summarizes the state of research, mechanisms studied in the lab, what data exist in humans, and the key gaps that must be addressed before any credible clinical use can be recommended.
BPC-157 is a synthetic peptide fragment originally identified in gastric juice. In laboratory research, it’s been studied for effects on connective tissue healing, angiogenesis (new blood vessel formation), and cellular repair processes. It has been proposed to activate molecular pathways involved in wound repair and cell survival.
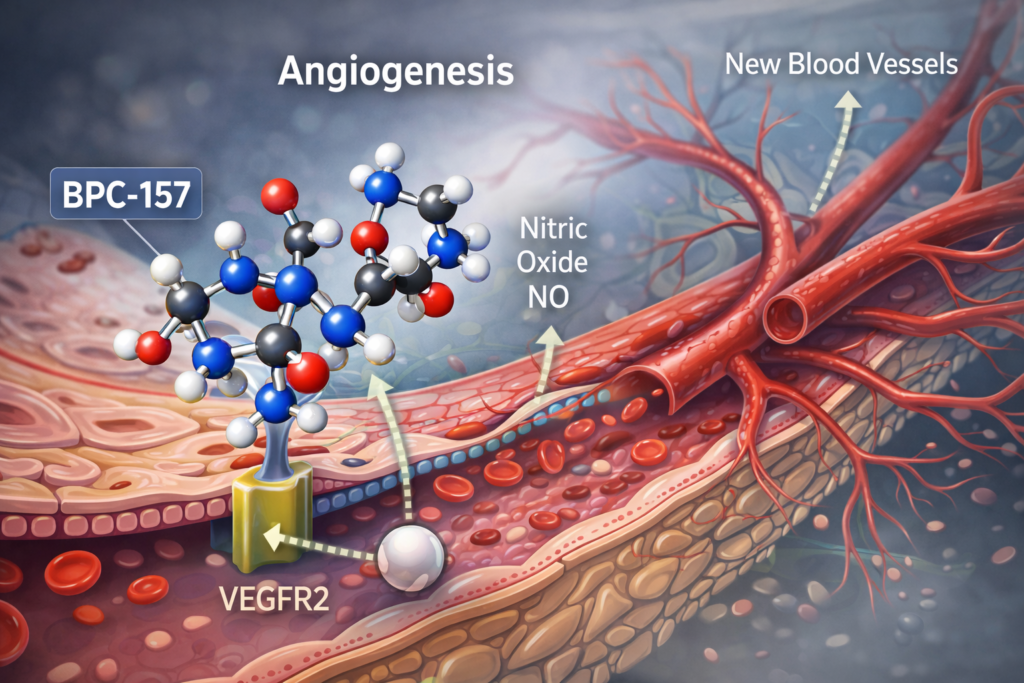
Laboratory and animal studies suggest that BPC-157:
These findings highlight biological activity, but do not confirm clinical safety or effectiveness in humans.
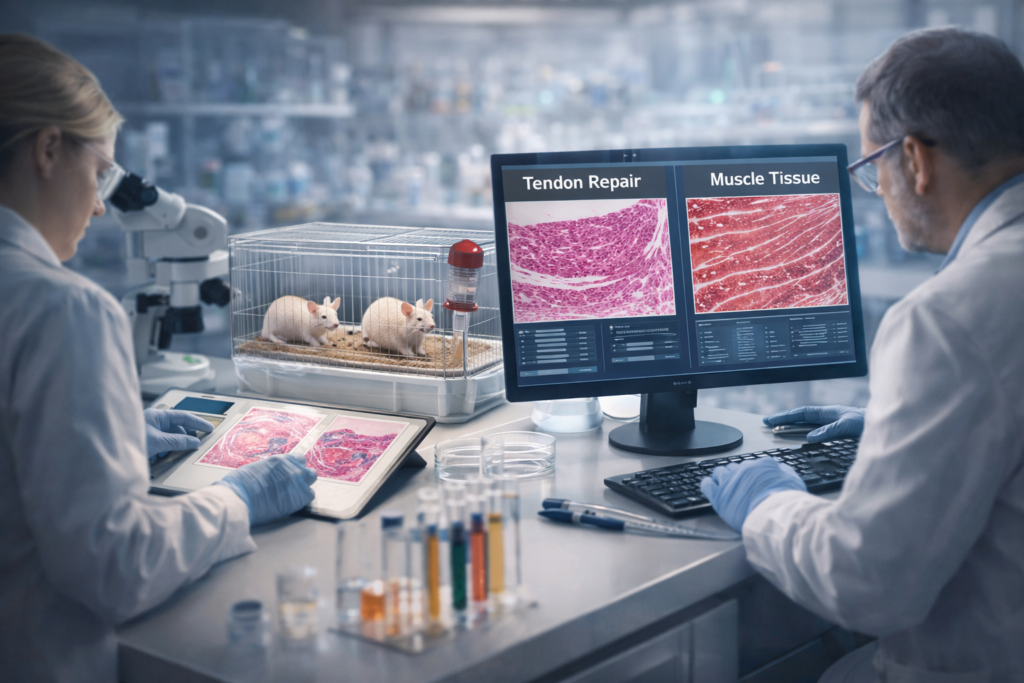
Across multiple rodent and in vitro studies, researchers have reported positive outcomes in models of:
Tendon and ligament injury
Muscle damage
Bone fracture models
Neurological injury and spinal cord trauma
Vascular occlusion syndromes
These preclinical results have motivated further research interest, particularly in regenerative medicine and orthobiologics.
Despite robust animal data, high-quality human clinical trials are sparse:
A retrospective observational report described relief in knee pain following intra-articular peptide injection in a very small group of patients, but lacked rigorous outcome measures.
A pilot safety infusion study reported no adverse lab values in two adults given BPC-157 intravenously, but this small sample cannot establish safety or efficacy.
A Phase I human trial registered to assess safety and pharmacokinetics was started and then cancelled without published results.
The majority of documented research is animal-based, and human outcomes remain virtually untested in controlled settings.
Several regulatory bodies have weighed in:
World Anti-Doping Agency (WADA) includes BPC-157 on its prohibited list because of performance-enhancing concern and lack of approved medical uses.
U.S. Food and Drug Administration (FDA) has not approved it for any medical indication, and many products marketed to consumers are sold as “research chemicals” without verified quality, purity, or standardized dosing.
Safety concerns include:
Unknown long-term effects in humans
Potential angiogenesis risk, which theoretically could affect tumor growth if present — a concern seen with other pro-vascular pathways in cancer biology
Contaminants or dosing variability in unregulated products
Without phase II/III clinical trials, there’s no reliable safety profile or established therapeutic utility.
While some providers and athletes share anecdotal reports of benefit, mainstream medical and research communities caution against clinical use without strong evidence. The surge in promotional marketing through influencers and wellness clinics often exceeds what current science supports.
For BPC-157 to be credible in medicine, the following are essential:
Controlled clinical trials assessing safety and efficacy across indications
Standardized peptide formulations with verified purity
Pharmacokinetic and dose-finding studies in humans
Long-term safety monitoring, including cancer risk and systemic effects
BPC-157 remains an intriguing biological molecule with promising preclinical results, especially in tissue repair and vascular modulation. But current research is dominated by animal and in vitro studies; human clinical data is extremely limited, making safety and efficacy unproven. Public use of BPC-157 falls outside regulatory approval, and experts advise prudence and further systematic investigation before clinical adoption.
References